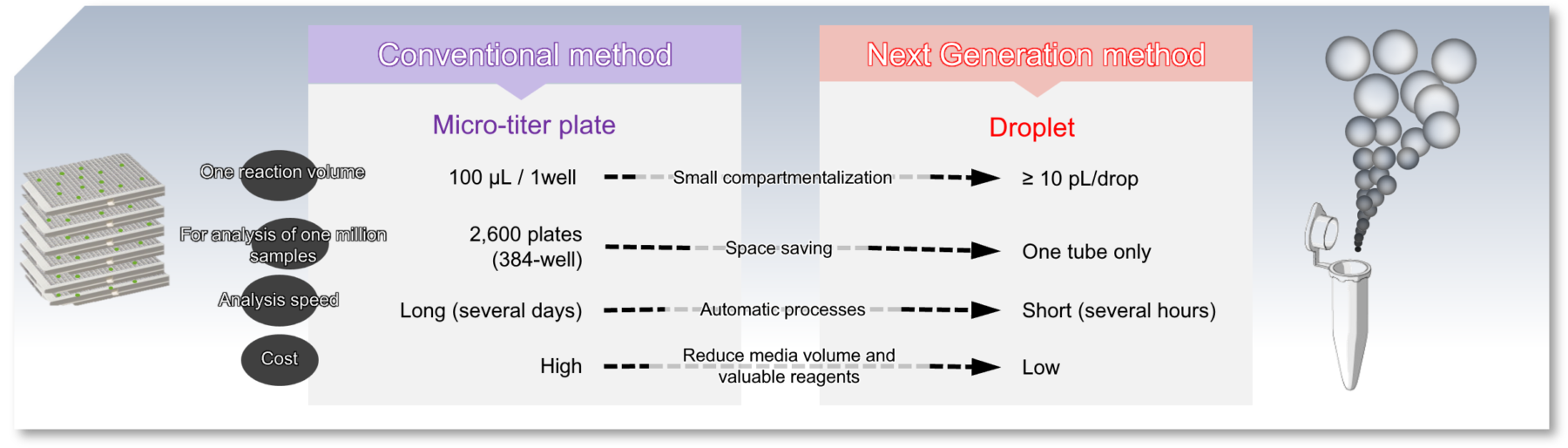
Droplets Applications
About Droplet

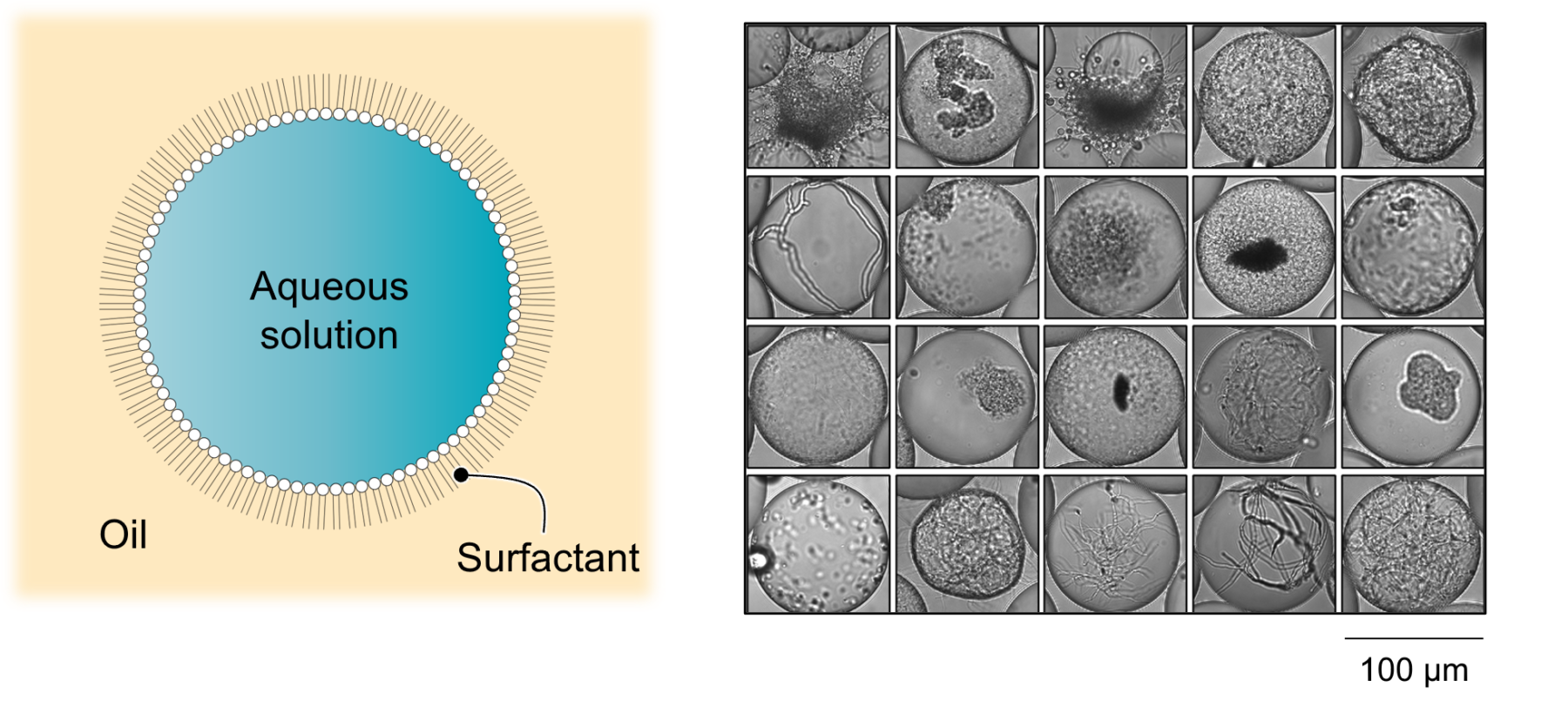
Microscopy of droplets
“Droplets” can be easily generated in large quantities and have the ability to encapsulate microorganisms or cells individually, allowing for the analysis of a wide variety of samples simultaneously and in parallel. Therefore, Droplet has been used in various fields, including high-throughput screening (HTS), making use of various advantages such as accelerated generation and analysis, small-bioreactor culture, and single-cell analysis.
Advantages of droplet technology
◾️Each droplet (picoliter-scale) functions as each well (microliter-scale) of a well-plate.
◾️Over one million droplets (reaction compartments) can be stored in a single tube.
◾️Significant savings can be achieved in analysis time, reagent consumption, and incubation space.
◾️Target droplet can be screened by On-chip Sort/On-chip Droplet Selector
Water-in-oil (W/O) droplets
Schematic of a W/O droplet (left) and microscopic image of environmental microorganism culture (right)
・Structure: Emulsion in which water droplets (e.g. the medium) are dispersed in oil and stabilized by surfactants.
・Features: Microorganisms/cells grow and metabolites accumulate in each droplet as they are compartmentalized by oil. The addition of substrates allows for the detection of metabolites along with the cultivation.
・Use: Microbial culture, screening of environmental microbes/mutant strains, directed evolution of enzymes, etc.
Single-cell cultivation
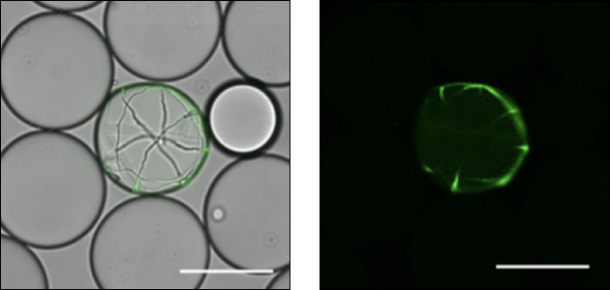
Filamentous fungi Trichoderma reesei expressing GFP Trichoderma reesei
Single-cell encapsulation and cultivation allow massively parallel screening based on the growth, protein expression, and secretion amount.
Luu, X. C., Shida, Y., Suzuki, Y., Sato, N., Nakamura, A., Ogasawara, W.. New Biotechnology 2022 72, 149-158.
Novel microorganisms screening

Microorganisms from industrial wastewater with specific protease activity
As a tool for isolating novel microorganisms from diverse environments, including soil, hydrosphere, and extreme environments.
Nakamura, A., Honma, N., Tanaka, Y., Suzuki, Y., Shida, Y., Tsuda, Y., Hidaka, K., Ogasawara, W., Analytical Chemistry 2022 94 (5), 2416-2424.
Cell-cell interaction
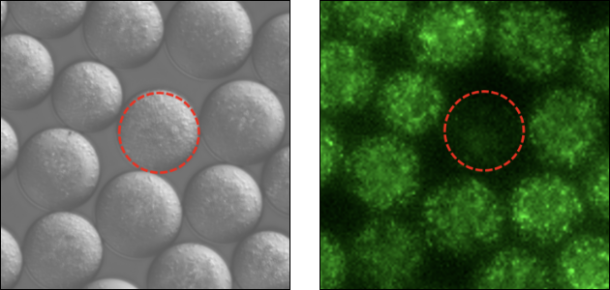
Co-encapsulation of GFP-expressing pathogens and soil microorganisms. Green fluorescence is attenuated in droplets showing antagonistic effects.
Droplet Library can be easily prepared containing combinations of various types of bacteria by taking advantage of the feature of high-throughput droplet generation.
The results are obtained in conjunction with Dr. Yasunori Ichihashi and Dr. Megumi Narukawa, RIKEN BioResource Research Center
Gel microdrops (GMD)
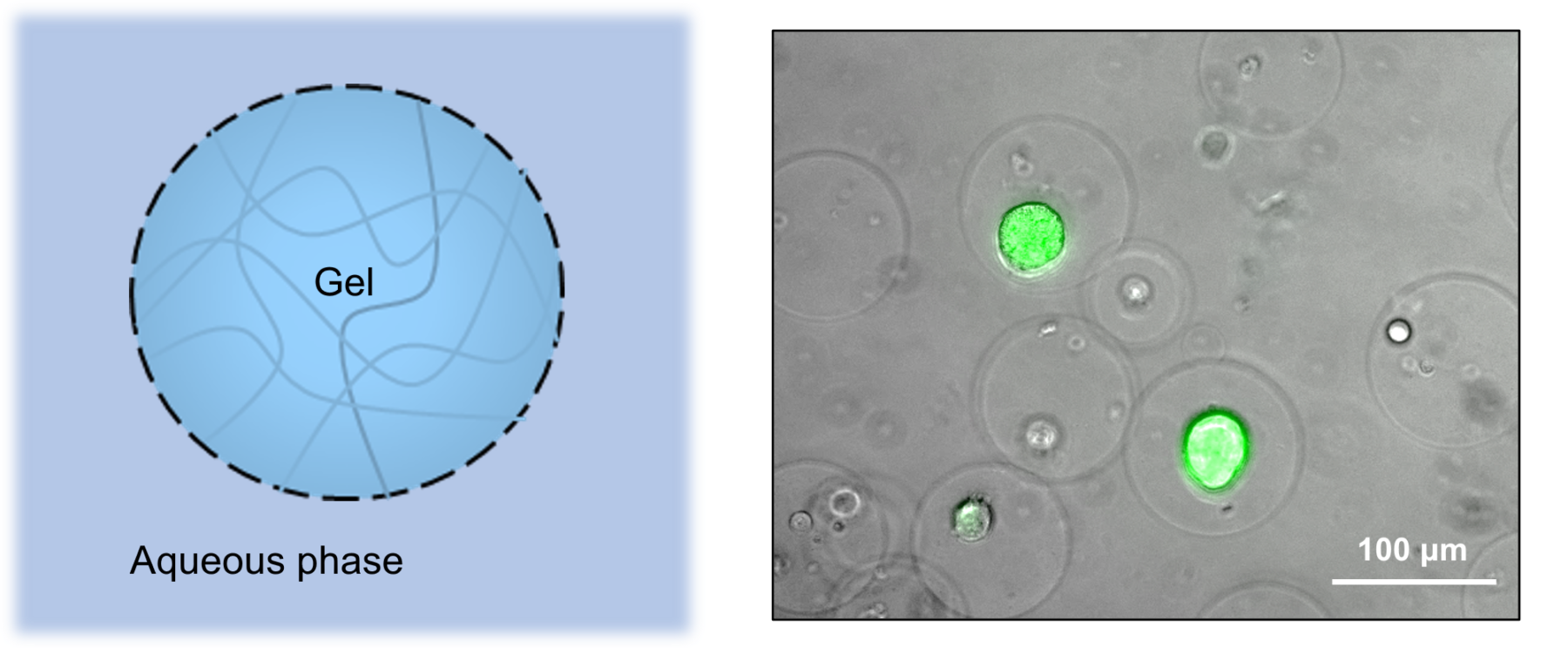
Schematic of a GMD (left) and microscopic image of cancer cell culture (right)
・Structure: Droplets with gelled interior.
・Features: Substances are highly permeable both inside and outside the droplet structure because the droplets can be dispersed in an outer aqueous phase, such as a culture medium, rather than in oil, while relatively large materials such as cells, beads, and genomes are retained by the gel as a scaffold.
・Use: Cell culture, screening of mutant strains/antibody-producing cells, microbial interaction studies, etc.
Isolation of Protein High-Producing Strains
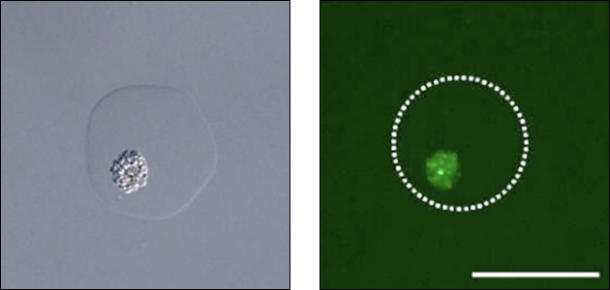
Recombinant protein-producing budding yeast
High-throughput screening of high-producing strains from a large number of candidates such as UV mutant libraries
Fujitani, H., Tsuda, S., Ishii, T., Machida, M., bioRxiv 2019
Screening based on substance production
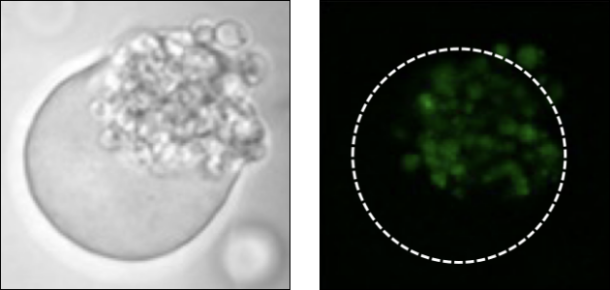
Oleaginous microorganisms stained with BODIPY
Staining of microorganisms after GMD cultivation allows screening based on substance production such as lipids.
Tanaka, Y., Nakamura, A., Suzuki, Y., Maruta, K., Shida, Y., Ogasawara, W. , Journal of Biotechnology 2022 358, 46-54.
Elucidation of the gut microbiota function
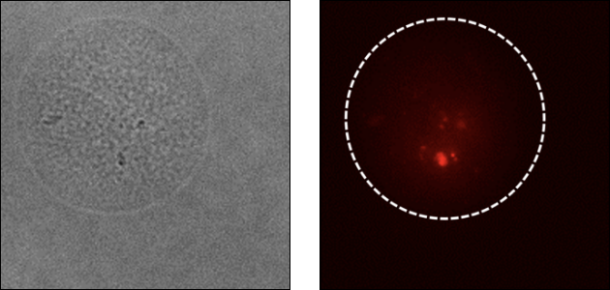
Elucidation of the gut microbiota function
High-throughput screening to elucidate the function of diverse gut microbiota
The results are obtained in conjunction with Prof. Shinji Fukuda, Institute for Advanced Biosciences, Keio University
Droplet Sorting by Microfluidics Chip
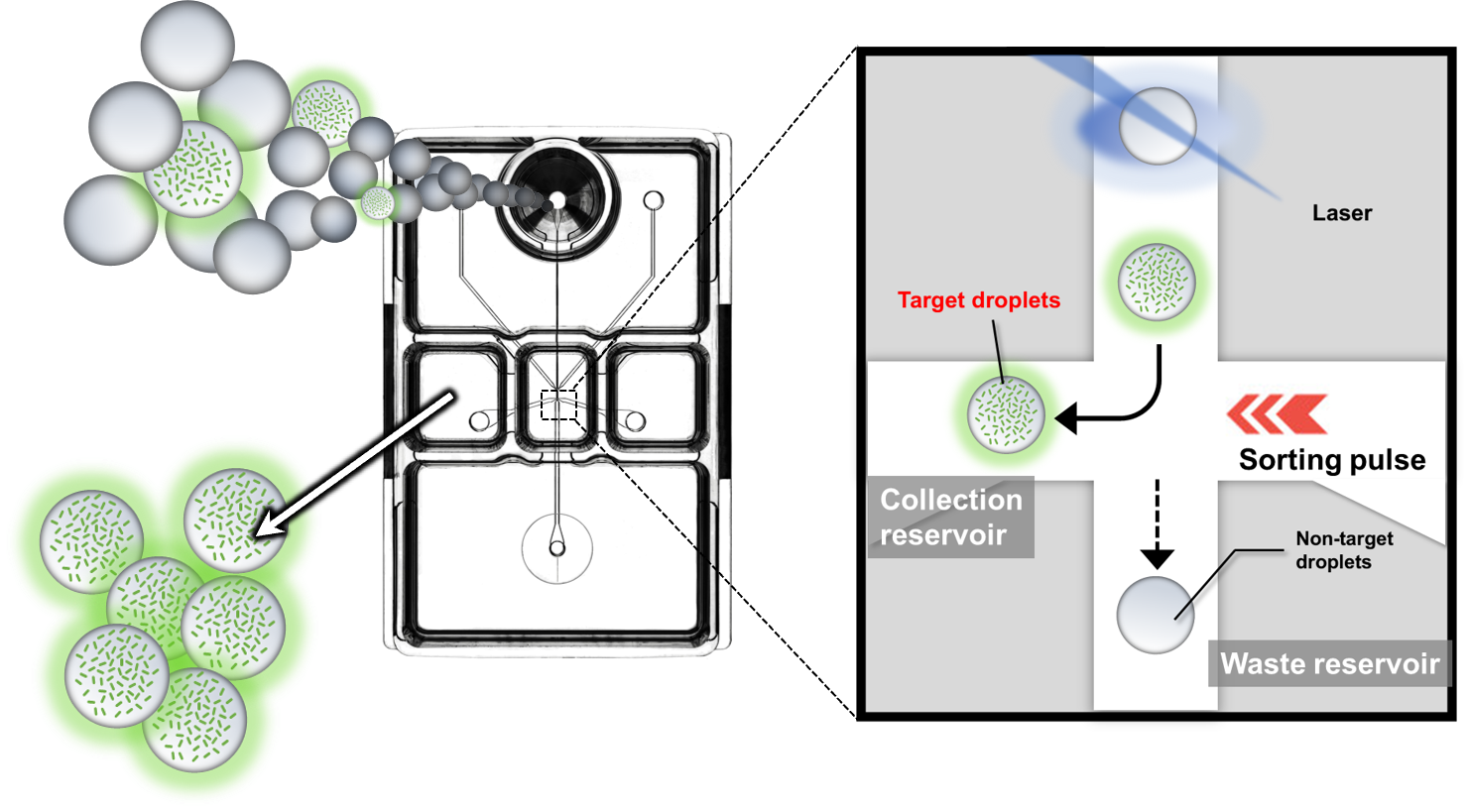
On-chip Sort / On-chip Droplet Selector enables the screening of droplets of interest based on enzyme reaction, proliferative activity, antibody reaction, etc.
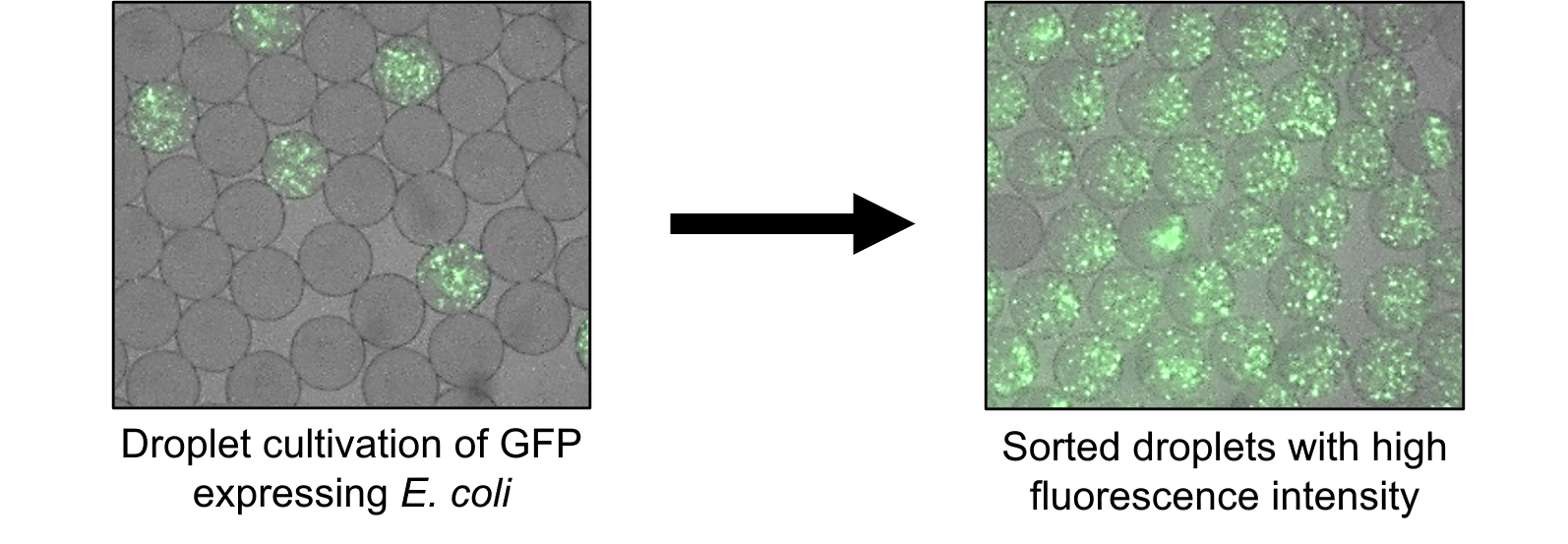
Detection and Screening of Droplets Encapsulating Microorganism
A major challenge in obtaining pure cultures of environmental microorganisms has been detecting and isolating droplets containing microorganisms with various growth rates from millions of droplets. To solve this problem, Dr. Noda and his team at the National Institute of Advanced Industrial Science and Technology (AIST) developed FNAP-sort (fluorescent nucleic acid probe in droplets for bacterial sorting), a method that utilizes fluorescence to detect droplets containing grown microorganisms. In this method, a fluorescent nucleic acid probe, which is quenched by FRET (fluorescence resonance energy transfer), is cleaved by RNase generated during microbial growth, resulting in the elimination of FRET and an increase in fluorescence emission.
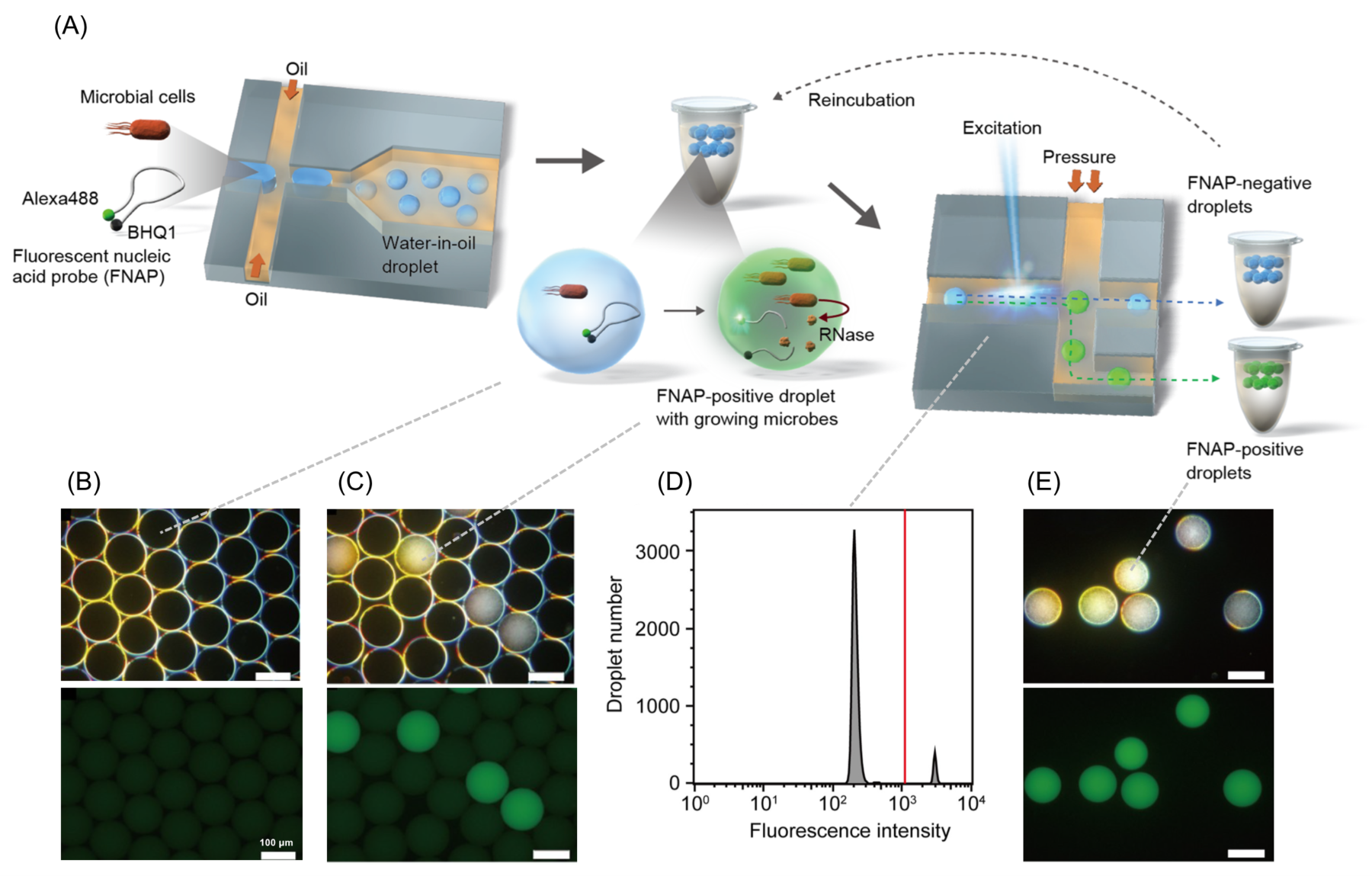
FNAP-sort workflow and proof of principle.
(A) FNAP-sort workflow, (B) microscopic images of droplets immediately after generation, (C) microscopic images of droplets after one day of incubation, (D) fluorescence histogram of droplets after one day of incubation, and (E) microscopic images of sorted droplets. Upper panels: dark-field observation; lower panels: fluorescence observation for (B), (C), and (E).
Saito, K., Ota, Y., Tourlousse, D. M., Matsukura, S., Fujitani, H., Morita, M., Tsuneda, S., Noda, N., Scientific Report 2021 11(1), 9506.
Products

“On-chip Droplet Generator”
Stable and uniform production of emulsion droplets. Using our unique microfluidic chip, stable and uniform droplets can be produced.

“On-chip® Droplet Selector”
Cell/microorganism screening at 1,000 times higher throughput than conventional screening methods.